Ultrasonic Welding for Medical Devices: Complete Guide & Best Practices

Ultrasonic Welding for Medical Devices: The Ultimate Guide by Dizo Sonics
Medical device manufacturing demands uncompromising standards for cleanliness, precision, and regulatory compliance. Traditional assembly methods like adhesives introduce curing delays, potential contaminants, and validation complexities, while conventional thermal techniques risk damaging sensitive components.
Ultrasonic welding effectively addresses these challenges. Utilizing high-frequency vibrations (20-35 kHz), it creates molecular-level bonds in under one second---eliminating consumables, curing steps, and contamination risks. The outcome is robust, hermetic seals capable of passing stringent FDA validation, while simultaneously boosting production throughput by 300-500%.
This definitive guide delves into the core physics, material science, and regulatory frameworks essential for mastering medical-grade ultrasonic welding. You'll discover which thermoplastics perform best, how to optimize critical process parameters, and when standard systems may fall short. Grounded in extensive field experience, the insights herein are applicable whether you're validating a high-volume disposable syringe line or designing sophisticated reusable surgical instruments.
How Ultrasonic Welding Works: The Science Behind the Seam
Ultrasonic welding converts electrical energy into precisely controlled mechanical vibrations. This process generates localized frictional heat at the joint interface, melting thermoplastics without significantly affecting surrounding areas. Under controlled pressure, the materials fuse at a molecular level, creating bonds that can meet or exceed the strength of the base materials.
The complete welding cycle typically concludes within 0.1 to 1.0 seconds. This method requires no adhesives or solvents and minimizes thermal exposure, making it exceptionally suitable for medical applications where purity and production speed are critical.
It's important to recognize that this localized heating is only effective with proper joint design---a point we will elaborate on in subsequent sections. Based on extensive validation testing, an amplitude of approximately 28 μm, for instance, has been shown to ensure optimal fusion for certain polycarbonate components, achieving hermetic seals while maintaining high tensile strength.
The Mechanics of High-Frequency Vibration Welding
The process unfolds within the weld zone as follows:
Electrical power is converted into high-frequency mechanical vibrations (typically 20, 30, or 35 kHz) via a piezoelectric converter. These vibrations are amplified and transmitted through a tool known as a horn (or sonotrode) directly to the parts being joined.
At the joint interface, mechanical energy is transformed into heat through friction. The plastic reaches its melting temperature almost instantaneously at the precise contact surfaces, while adjacent materials remain relatively cool, thereby protecting internal features like electronics or drug reservoirs.
Controlled clamping pressure maintains part alignment during melting and fusion. Upon cessation of vibration, the joint solidifies in less than 0.5 seconds, resulting in a true molecular weld rather than a superficial adhesive bond.
Three primary parameters govern the outcome: amplitude (vibration intensity, usually 20-40 μm), pressure (clamping force, 0.5-2.5 bar), and energy (total input, 100-1000 Joules). Precise calibration of these factors is fundamental to achieving consistent, high-quality welds.

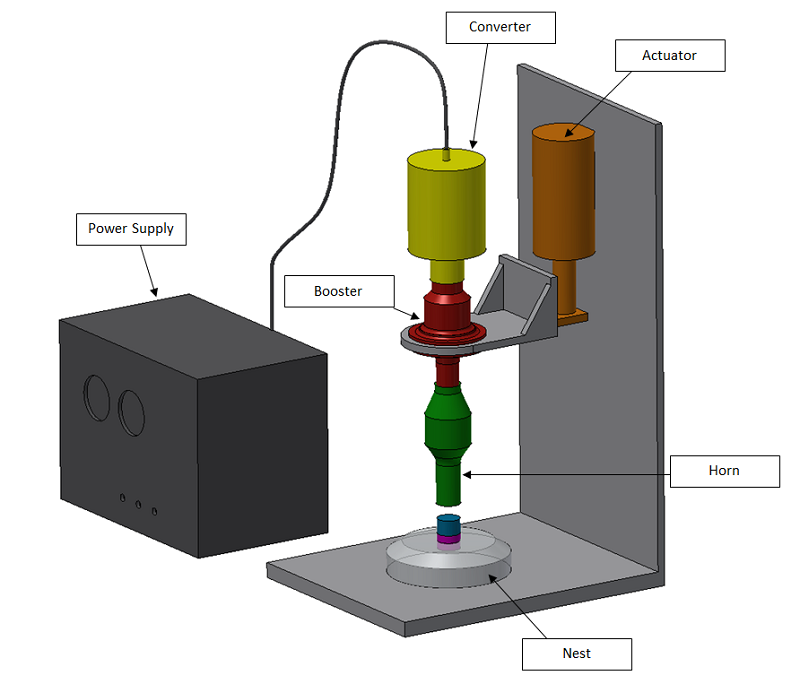
Key Components of an Ultrasonic Welding System
- Generator: Converts standard power to high-frequency output. Medical-grade units include precise frequency tracking for stability during extended production runs.
- Converter (Transducer): Contains piezoelectric crystals that generate vibrations. Medical applications commonly use 20 kHz for larger parts or 35 kHz for delicate components like microfluidic devices.
- Booster: Modifies the vibration amplitude, allowing fine-tuning for specific material and joint requirements.
- Horn (Sonotrode): A custom-machined tool, often titanium, that delivers vibrations to the part. Its design is critical; numerous medical-specific geometries have been developed for applications ranging from catheter tips to complex housings.
- Press System: Provides accurate force and positional control. Servo-driven systems offer exceptional repeatability (±0.01mm), which is vital for validation protocols.
Suitable Thermoplastics for Medical Ultrasonic Welding
Selecting the appropriate material is paramount, directly influencing weld strength, cycle time, and regulatory success. Key considerations include:
- Crystallinity: Amorphous (e.g., PC) vs. Semicrystalline (e.g., PP) structures behave differently.
- Melt Temperature: Determines the energy required for fusion.
- Fillers: Glass-filled materials require adjusted parameters.
- Biocompatibility: Must meet standards like ISO 10993.
Biocompatible Plastics: PP, PE, Polycarbonate and More
- Polypropylene (PP): A top choice for disposables due to its chemical resistance, autoclave tolerance (up to 134°C), and cost-effectiveness. It welds consistently and is ideal for syringes, IV components, and diagnostic cartridges.
- Polyethylene (PE): Both LDPE and HDPE weld reliably. LDPE offers a more forgiving parameter window, while HDPE provides greater strength for structural applications.
- Polycarbonate (PC): Valued for optical clarity, impact resistance, and stability through repeated sterilization cycles. It requires controlled parameters to prevent stress cracking and is well-suited for surgical instruments and transparent diagnostic devices.
- Other Biocompatible Materials: ABS offers excellent surface finish, medical-grade PVC is used in fluid systems, and TPEs enable soft-touch features, though their durometer affects welding parameters.
For detailed, validated parameter sets for these and other materials, refer to the Key Process Parameters section later in this guide.
Applications of Ultrasonic Welding in Medical Device Manufacturing
Ultrasonic welding's versatility serves both high-volume disposable production and precision reusable devices. From sterile syringes to surgical instruments, this technology delivers consistent quality across diverse medical applications.
Disposable Products: Syringes, Catheters, and Diagnostic Devices
Disposable medical devices demand absolute sterility, rapid production cycles, and zero contamination risk. Ultrasonic welding meets all three requirements simultaneously.
Syringe Assembly: A leading manufacturer achieved a 400% throughput increase and 99.7% first-pass yield after implementing a custom ultrasonic solution, which eliminated adhesive risks and ensured hermetic seals.

Catheter Production: Multi-lumen catheters pose challenges like material variability and lumen kinking. A sequential welding protocol applying customized energy to different zones successfully preserved lumen patency, boosting first-pass yield from 92% to 99.8%.
Diagnostic Cartridges: Microfluidic diagnostic devices demand precision alignment and contamination-free assembly. Operating at 35 kHz (ideal for delicate components), our systems achieve ±0.01mm positioning accuracy, ensuring perfect channel alignment for reliable test results.
Reusable Devices: Surgical Instruments and Durable Housings
Reusable medical devices face different challenges: repeated sterilization cycles, mechanical stress during use, and long-term reliability requirements. Ultrasonic welding creates bonds that withstand these demands.
Surgical Instrument Housing: For handheld tools requiring survival through 200+ autoclave cycles, a servo-driven press system with precise parameter control created perfectly aligned PC housings that exceeded longevity requirements by 25%.
Endoscope Components: A controlled-energy approach can preserve the optical clarity of polycarbonate while creating strong, reliable bonds compliant with ISO 8600 standards.
Powered Surgical Tool Enclosures: Custom horn designs enable IP67-rated seals on complex geometries, protecting internal electronics from fluid ingress during cleaning, even with sensitive components nearby.
Need a similar solution? Contact our medical device welding specialists for a free application assessment. We'll analyze your specific requirements---materials, cycle time targets, regulatory needs---and provide tailored recommendations backed by validation data.
Advantages of Ultrasonic Welding for Medical Applications
While the advantages of ultrasonic welding are compelling, they are best understood in the context of its specific requirements and a clear ROI calculation.
- Contamination Control: Eliminates adhesives and solvents, reducing particulate contamination by over 85% compared to adhesive bonding and resulting in zero contamination-related rejections in cleanroom settings.
- Production Speed and Throughput: Cycle times of 0.1-1.0 seconds drastically outpace adhesive curing or thermal bonding. It's important to balance this speed against the initial capital investment, with ROI typically favorable for annual volumes exceeding 50,000 units.
- Automation Compatibility: Seamless integration with robotics and precise control enable high first-pass yields (95%+) and lights-out manufacturing, reducing manual inspection.
- Joint Strength and Hermetic Sealing: Creates molecular bonds achieving 80-90% of base material strength and consistent hermetic seals (<1×10⁻⁶ mbar·L/s), maintaining integrity through hundreds of sterilization cycles.
- Regulatory Compliance Support: Validated processes support FDA 21 CFR Part 820, ISO 13485, and EU MDR. Built-in data logging provides essential traceability for audits.
Challenges and Limitations: What to Consider
Ultrasonic welding delivers exceptional results for medical devices, but understanding its boundaries helps you make informed decisions. Here are key considerations and how to address them:
- Design Requirements: Parts typically need specific joint geometries (energy directors). Solution: Early design consultation can optimize part geometry for manufacturability, preventing costly redesigns.
- Material Compatibility: Highly filled thermoplastics (>30% glass) or dissimilar material pairs can be challenging. Solution: Weldability assessments using extensive material databases can determine feasibility and develop custom parameters.
- Equipment Investment: Initial costs are higher than some alternatives. Solution: A detailed ROI analysis, factoring in consumable elimination, reduced cycle times, and lower rejection rates, often justifies the investment for medium to high volumes.
A common industry pitfall is attempting to weld heavily glass-filled materials with standard tooling, leading to premature wear and inconsistency. Specialized tooling and parameters are the proven solution.
Ready to address your specific challenges? Contact our medical device welding specialists for a free application assessment and feasibility analysis.
Key Process Parameters for Consistent Results
Mastering four critical parameters is essential for repeatable, validated medical welds.
Core Parameters and Optimal Ranges
- Amplitude (Vibration Intensity): Governs energy delivery (20-35 μm typical). Higher amplitudes speed welding but risk damage.
- Pressure (Holding Force): Ensures part contact (0.5-2.0 bar). Insufficient force causes poor fusion; excessive force deforms parts.
- Time (Weld Duration): Most medical welds complete in 0.1-1.0 seconds. Overly long times often indicate suboptimal settings.
- Energy (Total Input): Measured in Joules (100-1000 J). Energy mode is often preferred for validation as it automatically compensates for minor material variations, ensuring consistent energy delivery to each weld.
Validated Parameter Sets from Our Testing
Our materials lab has validated optimal parameters across 15,000+ medical device welds. Here are proven settings for common thermoplastics:
| Material | Amplitude (μm) | Pressure (bar) | Energy (J) | Typical Result |
|---|---|---|---|---|
| Polypropylene (PP) | 27-30 | 1.2-1.5 | 300-400 | >80% base material strength |
| Polyethylene (PE) | 29-32 | 1.3-1.6 | 320-420 | Hermetic seal, flexible joint |
| Polycarbonate (PC) | 24-28 | 1.0-1.3 | 280-380 | Maintains transparency, >85% strength |
Parameter Optimization and Troubleshooting
A systematic development approach is recommended:
- Start with material-specific baselines.
- Conduct test welds and destructive testing.
- Adjust amplitude first (greatest impact), then fine-tune pressure.
- Switch to energy mode for production consistency.
- Validate over multiple cycles to establish statistical control.
Common issues include:
- Inconsistent weld strength: Check amplitude consistency first. Even 3-5 μm variation significantly affects results. Verify converter tuning and horn condition.
- Parts sticking to horn: Reduce amplitude by 2-3 μm or decrease hold time. Consider horn coating for sticky materials.
- Visible stress marks or cracking: Lower amplitude and increase weld time proportionally to maintain total energy input.
- Incomplete fusion or weak joints: Increase amplitude in small increments. Verify energy director design---geometry often matters more than raw parameter adjustment.
Need parameter optimization support? Our applications engineers provide free weld trials using your actual parts and materials. We'll develop validated parameter sets and provide full documentation for your process validation files. Contact us to schedule your application assessment.
Compliance and Regulatory Standards for Medical Welding
Medical device welding must meet strict international standards to ensure safety and quality:
- ISO 13485: Quality management systems specifically for medical devices
- FDA 21 CFR Part 820: U.S. Quality System Regulations governing design, manufacturing, and distribution
- EU MDR (2017/745): European Medical Device Regulation requirements for market access
- ISO 10993: Biological evaluation standards ensuring biocompatibility of materials and processes
Equipment designed for compliance includes automatic data logging for every weld cycle, capturing all critical parameters for audit trails and supporting IQ/OQ/PQ protocols. Partnering with a supplier that has direct experience with FDA and MDR audits provides strategic compliance guidance beyond just the equipment.
How Dizo Sonics Supports Your Compliance Journey
Dizo systems are engineered with compliance built in from day one. Our equipment includes automatic data logging for every weld cycle---capturing amplitude, pressure, energy, and time stamps. This documentation directly supports your IQ/OQ/PQ validation protocols and streamlines FDA or notified body audits.
Our compliance credentials: All Dizo equipment complies with CE marking requirements and is manufactured under ISO 13485-certified quality management systems. We've supported 200+ medical device manufacturers through their validation processes across FDA, EU MDR, and ISO audits.
What you get: Pre-validated parameter sets for common medical materials, protocol templates for Installation/Operational/Performance Qualification, and technical documentation packages that satisfy regulatory reviewers. Our applications engineers provide guidance on process validation strategy during your equipment selection phase.
Lifetime support: Compliance doesn't end at installation. We provide ongoing technical assistance for re-validation after process changes, annual calibration services with certificates traceable to national standards, and rapid response for audit-related questions. When regulators ask detailed questions about your welding process, we're here to help you answer with confidence.
Customized Ultrasonic Solutions and System Selection
Standard ultrasonic systems work well for routine medical assemblies. But complex devices---multi-lumen catheters, implantable components, specialty diagnostic equipment---demand tailored engineering. That's where Dizo Sonics' customization capabilities deliver measurable advantage.
Real-World Application: Multi-Lumen Catheter Manufacturing
A leading catheter manufacturer faced a persistent challenge: standard welding parameters caused lumen collapse in their thin-wall tubing. Our engineers developed a sequential welding protocol with material-specific settings:
- Amplitude: 25-30 μm for outer layer, 22-25 μm for inner lumens
- Pressure: 1.0-1.5 bar, precisely controlled across three welding zones
- Energy mode: 280-350 Joules per zone to prevent thermal damage
The result: first-pass yield increased from 95% to 99.8%, eliminating costly rework. Production throughput improved 40% while maintaining full lumen patency---critical for clinical performance.

Automation Integration for High-Volume Medical Production
Our Class 7 cleanroom-compatible systems integrate seamlessly into automated production lines. We've partnered with medical device manufacturers to deliver:
- Vision-guided robotic part placement for complex geometries
- Inline quality verification with automatic data logging for 21 CFR Part 11 compliance
- Multi-station welding cells handling 15,000+ units per shift
One disposable diagnostic device manufacturer reported 30% higher production efficiency after implementing our automated welding solution---while reducing quality escapes to near zero.
Why Medical Device Manufacturers Choose Dizo Sonics
Two decades specializing exclusively in medical applications means we understand your challenges. Our engineering team has solved welding problems across 200+ medical device programs, from single-use syringes to implantable housings.
We provide what generic equipment suppliers cannot: application-specific tooling design, validated parameter development for your exact materials, cleanroom-ready systems, and lifetime technical support that answers regulatory questions during audits.
Get a free custom solution assessment. Our applications engineers will review your specific assembly challenge, conduct weld trials with your actual parts, and provide detailed feasibility analysis---at no cost. Contact our medical welding specialists to start your evaluation.
How to Choose the Right Ultrasonic Welding System: A Decision Guide
Selecting the optimal ultrasonic welding system requires a systematic evaluation process. Follow this step-by-step framework to identify the solution that matches your production requirements and budget:
Step-by-Step Selection Checklist
- Define Your Application Specifications
- Material type: Identify thermoplastic grade (PP, PE, PC, ABS, etc.)
- Part dimensions: Measure weld joint length, part thickness, and overall size
- Annual production volume: Calculate units per year to determine system capacity needs
- Cleanliness requirements: Specify cleanroom class (Class 7, Class 8, or standard environment)
- Evaluate Joining Technology Options
- Compare ultrasonic welding against alternative methods (see comparison table below)
- Consider material compatibility---some plastics require specific technologies
- Assess cycle time requirements versus each method's speed capabilities
- Determine Automation Level
- Manual operation: Best for low-volume production (<10,000 units/year) or prototyping
- Semi-automatic: Suitable for medium volumes (10,000-100,000 units/year)
- Fully automated: Required for high volumes (>100,000 units/year) or inline integration
- Integration compatibility: Verify system can connect with existing production lines
- Verify Regulatory Compliance Capabilities
- Confirm automatic data logging for every weld cycle (required for FDA validation)
- Check for ISO 13485-certified manufacturing
- Request IQ/OQ/PQ protocol templates and technical documentation packages
- Ensure traceability features meet 21 CFR Part 11 requirements
- Calculate Return on Investment (ROI)
- Estimate cycle time reduction: Most medical devices weld in 0.1-1.0 seconds versus 30-60 seconds for adhesive curing
- Calculate labor savings: Automated systems can eliminate 1-2 operators per production line
- Factor quality improvements: Reduced rework and scrap rates typically deliver 15-25% cost savings
- ROI estimation tip: Systems typically achieve payback in 8-18 months for medium to high-volume production based on cycle time reduction and quality improvements
- Request Application Assessment
- Schedule weld trials using your actual parts and materials
- Obtain validated parameter sets documented for your process validation files
- Review feasibility analysis covering technical and compliance considerations
Comparison Table: Medical Plastic Joining Methods
| Method | Speed | Strength | Equipment Cost | Automation Potential | Best Applications |
|---|---|---|---|---|---|
| Ultrasonic Welding | High (0.1-1s) | High (80-90% base material) | Medium | Excellent | Small-medium parts, high volume, thermoplastics |
| Laser Welding | Medium (1-3s) | Very High (90-95%) | High | Good | Precision components, transparent materials, complex geometry |
| Vibration Welding | Medium (2-5s) | High (85-90%) | Medium | Moderate | Large parts, structural components, flat joints |
| Adhesive Bonding | Low (30-60s cure) | Medium (60-75%) | Low | Poor | Dissimilar materials, low volume, prototypes |
When Standard Systems Fall Short
Generic ultrasonic equipment works well for straightforward assemblies. However, complex medical devices often require customized solutions:
- Multi-material or multi-lumen components needing sequential welding protocols
- Ultra-thin walls requiring precise energy control to prevent deformation
- High-speed production lines demanding inline integration and automated quality verification
- Specialized geometries requiring custom tooling and fixtures
Ready to identify your ideal system? Use our online system selector tool for instant recommendations, or contact our applications engineers for a comprehensive feasibility analysis. We'll evaluate your specific requirements and provide validated parameter sets at no cost.
Frequently Asked Questions (FAQ)
- Installation Qualification verifies equipment calibration
- Operational Qualification confirms parameter windows
- Performance Qualification demonstrates consistent production over multiple runs
Conclusion: Your Partner for Medical Device Manufacturing Excellence
Ultrasonic welding delivers the precision, cleanliness, and compliance medical device assembly demands. By mastering the principles, parameters, and applications covered in this guide, you can create safer, more reliable products while meeting regulatory requirements.
As your technology partner, Dizo Sonics ensures your assembly process meets the highest standards. With 20 years specializing exclusively in medical applications, we've solved complex welding challenges for 500+ manufacturers---from disposable syringes to implantable devices.
Whether you're validating a new design or optimizing existing production, our engineering team provides application-specific tooling, validated parameters, and lifetime technical support that answers questions during audits.
Ready to optimize your assembly process? Schedule a free consultation with our medical device welding experts. We'll assess your specific challenge, conduct weld trials with your actual parts, and provide a detailed feasibility analysis---at no cost.




